

More Posts from
Gut Microbiome & Digestion

Meet Your Microbiome: The Human Microbiome and How to Optimize It
Meet Your Microbiome: The Human Microbiome and How to Optimize

Benefits of Probiotics Including Improved Gut Health
Benefits of Probiotics Including Improved Gut Health “Probiotics” has become

Gut Microbiome Health and the Gut-Immune System Connection: Part 2 Lower GI
Gut Microbiome Health and the Gut-Immune System Connection: Part 2
Many people are enjoying the benefits of MegaPrebiotic in their life. This post will go in depth as to what MegaPrebiotic is and how it works in the human body.
Background of MegaPrebiotic (and what are prebiotics?)
Prebiotics are non-digestible fibers that feed the bacteria living in your gut. However, most prebiotics on the market can feed both harmful and beneficial gut health bacteria, which can make some digestive issues worse. When it comes to the gut microbiome, a more diverse array of bacteria in the intestines lends itself to a much stronger and healthier gut – which means a stronger and healthier you! MegaPreBiotic is a cutting-edge Precision Prebiotic that can increase microbial diversity in the gut and selectively feed beneficial, keystone bacteria like Akkermansia muciniphila, Faecalibacterium prausnitzii, and Bifidobacterium spp.
MegaPrebiotic Ingredients
MegaPrebiotic is a Precision Prebiotic supplement made up of clinically-tested, non-digestible oligosaccharides. Oligosaccharides are essentially medium-chain carbohydrates. The prefix oligo- means few, which means that there are only a few links in the chain. Mono- and disaccharides have one or two links in their chains, respectively, and polysaccharides contain many links in the molecular chain. If carbohydrates cannot be digested and absorbed by the human intestinal tract, then they will pass down into the large intestine, where they function as fiber. The term fiber is generally used to refer to complex, long-chain carbohydrates that cannot be digested by our digestive enzymes or pancreatic juices, but it can also refer to a non-digestible carbohydrate. In this sense, non-digestible oligosaccharides can be considered functional fibers because they perform the same function as complex dietary fibers, and that function is to be fermented by intestinal bacteria.
When your gut health bacteria ferment fibers, they can produce gas, like methane, as a by-product or short-chain fatty acids, like butyrate, propionate, and acetate. Short-chain fatty acids are extremely important in the intestines because they are the prime energy source for your intestinal cells, known as enterocytes, but they can also soothe intestinal inflammation – which is the source of systemic inflammation. For this reason, a gut microbiome that produces a large amount of SCFAs will be much more capable of fighting inflammation and protecting the human host from disease.
The functional fibers used in MegaPreBiotic are fructo-oligosaccharides (FOS) from organic green and gold kiwifruit, xylo-oligosaccharides (XOS) from organic corn cob, and galacto-oligosaccharides (GOS) from lactose, derived from rBST-free cow’s milk.
[optin-monster-shortcode id=”ix4s9xeikqzz70yhc1ub”]
While the thought of corn and cow’s milk may cause some to back away in fear, MegaPreBiotic is well-tolerated by those with corn and dairy sensitivities. The reason for this is that most food allergies and sensitivities are adverse reactions to one of the proteins in these foods. For example, dairy sensitivities are often a reaction to casein, whey, or beta-lactoglobulin, while a dairy intolerance is the inability to properly digest lactose. MegaPreBiotic only contains oligosaccharides from the aforementioned sources, so there are no offending proteins or disaccharides in the formula
MegaPreBiotic is a powder lightly sweetened with stevia and monk fruit extracts with an organic mango flavor. While it may not make immediate sense, organic and natural mango flavoring does not actually contain any mango, so it should be safe to use if you have a mango allergy. The reason for this has to do with flavoring and food science. Extracts taken directly from mango will not necessarily taste like mango to your brain, so flavoring experts use other natural food components to create a flavor profile that tastes like mango to your taste buds and your brain.
MegaPreBiotic Function
Keystone bacteria like Akkermansia muciniphila, Faecalibacterium prausnitzii, and Bifidobacterium species are very important for human health, but their populations can be easily diminished by antibiotics, stress, diet, glyphosate, and other environmental toxins.
Akkermansia muciniphila is a next-generation beneficial microbe that affects glucose metabolism, lipid metabolism, and intestinal immunity.2 One study exploring the association between A. muciniphila and host characteristics found that at baseline, A. muciniphila was inversely related to fasting blood glucose levels, waist-to-hip ratio, and subcutaneous fat.3 Subjects with higher abundance of A. muciniphila exhibited the healthiest metabolic status, particularly in fasting blood glucose, triglycerides, and body fat distribution. They also displayed greater improvements in blood sugar regulation and other clinical parameters after calorie restriction, suggesting that A. muciniphila is associated with a healthier metabolism. Additionally, Amuc-1100, an outer membrane protein of Akkermansia has been identified as a compound that can enhance the intestinal barrier and induce the production of the anti-inflammatory cytokine interleukin-10 (IL-10).2 Low levels of A. muciniphila have been associated with obesity, diabetes, liver disease, cardiometabolic diseases, and low-grade inflammation.
Faecalibacterium prausnitzii is a butyrate-producing bacterium belonging to the Firmicutes phylum that is one of the most abundant and important commensal organisms in the human gut. By producing butyrate, F. prausnitzii plays a significant role in providing energy for intestinal cells and reducing intestinal inflammation. Low levels of F. prausnitzii are common signatures of inflammatory intestinal disorders like IBS, Crohn’s disease, ulcerative colitis, colorectal cancer, obesity, and celiac disease.4
Gut bacteria that fall under the genus of Bifidobacterium also play an important role in maintaining barrier function and stimulating the immune system.5 Because Bifidobacteria are also butyrate-producers, they can help reduce intestinal inflammation and also appear to help with weight management.6 Low levels of Bifidobacteria have been associated with obesity, diabetes, celiac disease, allergic asthma, dermatitis, IBD, chronic fatigue syndrome, and psoriasis.7-9
Given the laundry list of health benefits, the natural conclusion is that we should attempt to reseed the gut with these bacteria by taking them orally as probiotics. However, these anaerobic bacteria require oxygen-free environments in order to thrive and are not designed by nature to be reintroduced to the gut orally. Additionally, the aerobic environments in most manufacturing plants make it extremely difficult to produce truly anaerobic probiotics.
The good news is that these keystone strains are already present in the gut microbiome. Antibiotics, diet, stress, and other factors may reduce their abundance, but these commensal bacteria are never completely eradicated – they simply need to be revitalized. Just like a garden needs continuous care and proper fertilization, a healthy gut microbiome needs to be nurtured and reinforced with a Precision Prebiotic.
MegaPreBiotic Research
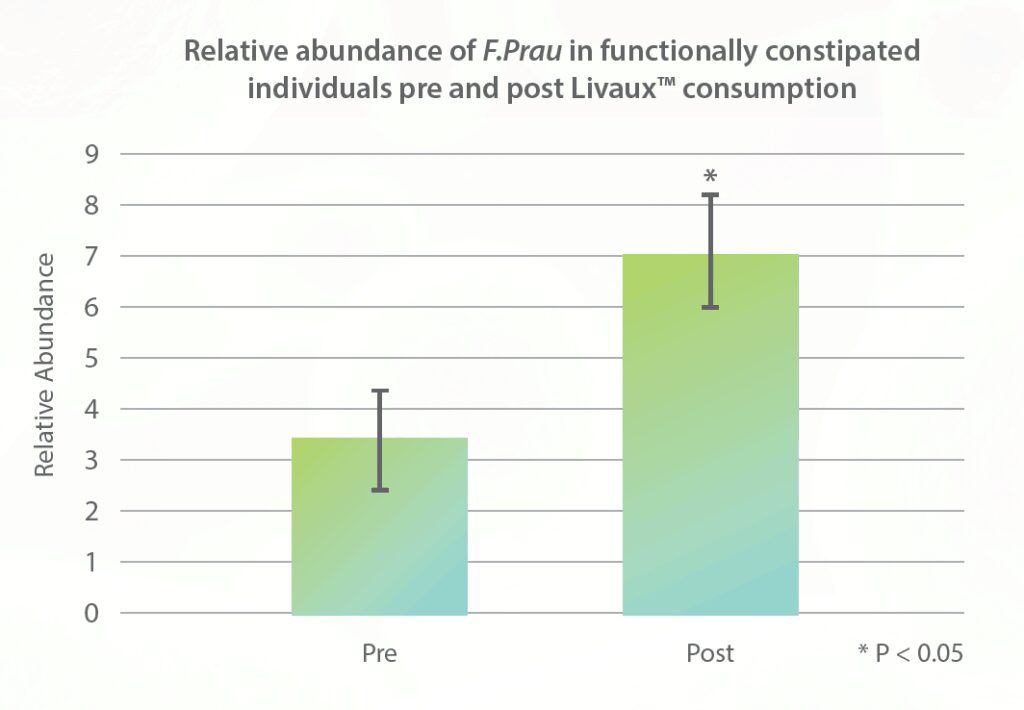
The fructooligosaccharides used in MegaPreBiotic™, ACTAZIN™ and Livaux™, were investigated in a randomized, double-blind, and placebo-controlled crossover trial in New Zealand assessing their prebiotic activity over the course of 6 months. This 2017 study, published in the Journal of Nutritional Sciences, found that supplementation with 2.4 g/day of Livaux™ increased the abundance of Faecalibacterium prausnitzii by 100% in as little as 4 weeks (see figure below).10
Another study published in Diabetes found that FOS supplementation was correlated with an 8,000% increase in A. muciniphila in only 5 weeks.11
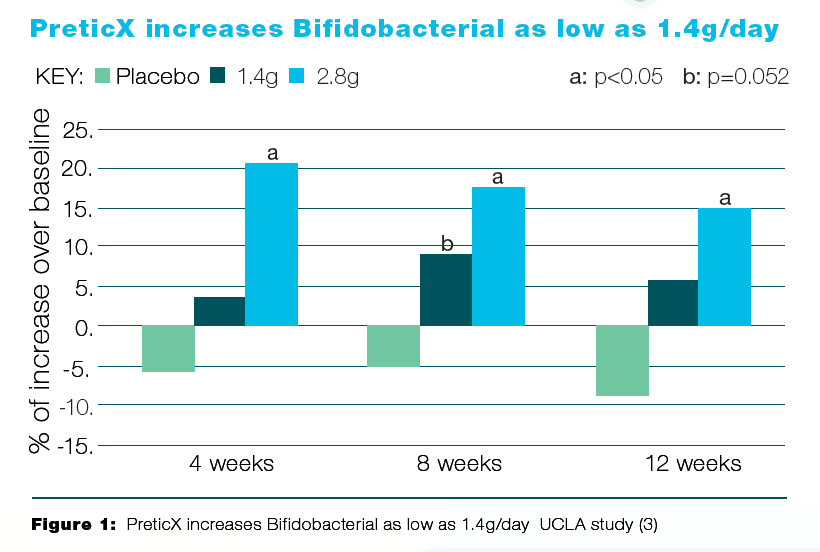
In 2014, a double-blind, randomized, and placebo-controlled study conducted at the University of California in Los Angeles was published in the Food and Function journal evaluating the efficacy and dosing of PreticX, the xylooligosaccharide ingredient used in MegaPreBiotic.12 Thirty-two healthy adult Subjects received 1.4 g PreticX, 2.8 g PreticX, or placebo in daily doses. The study consisted of a 2-week run-in, an 8-week intervention, and a 2-week washout phase. In both groups receiving PreticX, the abundance of Bifidobacterium spp increased. In the group receiving the larger dose, the abundance of Bifidobacterium increased by 21% in 4 weeks (See Figure 1 below).
In 2008, a randomized, double-blind, and placebo-controlled crossover study was published in the American Journal of Clinical Nutrition evaluating the prebiotic potential of Bimuno® galactooligosaccharides – the same ingredient used in MegaPreBiotic. This study included 59 healthy adult subjects, who received either 0, 3.6, or 7 grams of GOS for a 7-day treatment period with a 7-day washout period. The study found that Bimuno® GOS, derived from lactose, increased the abundance of Bifidobacterium by 66% in just 7 days.13
Furthermore, Bimuno® GOS (B-GOS®) has been shown to reduce the physical effects of digestive discomforts like bloating, abdominal pain, and gas.14-16 People who present with these symptoms are often diagnosed with Irritable Bowel Syndrome (IBS) and advised to avoid easily fermentable foods by following a low FODMAP diet, which stands for Fermentable Oligo-, Di-, Monosaccharides and Polyols. However, these foods are easily fermentable because they have prebiotic capabilities, and not all prebiotics are the same.
Although GOS falls under the oligosaccharide category in the low FODMAP diet, B-GOS® has been shown to reduce digestive symptoms as effectively as a low FODMAP diet while simultaneously increasing the abundance of Bifidobacterium species.16 Another proof-of-concept study noted that the gut microbiome has an adaptive response to B-GOS®. Those who experienced gas when starting supplementation with B-GOS® saw a resolution of symptoms by day 10.17
MegaSporeBiotic + MegaPreBiotic Synbiotic study
In 2018, MegaSporeBiotic and MegaPreBiotic were used in a gut model study using SHIME® technology, or the Simulator of the Human Intestinal Microbial Ecosystem.
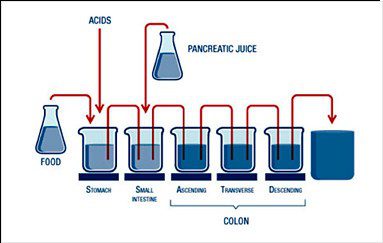
The SHIME® is a unique scientifically-validated, dynamic model of the complete gastrointestinal tract from the stomach to the colon used to study chemical, enzymatic, and microbial variables of the digestive tract in a controlled in vitro setting. This model includes 5 reactors that simulate the stomach, small intestine, and the 3 regions of the large intestine: the ascending, transverse, and descending colon. Careful control of the environmental variables in each reactor allows companies to study the effects of a compound or finished product in each segment of the digestive tract as well as the gut microbiome over several weeks.
The majority of gut bacteria can be found floating around in the intestinal lumen, but a small fraction of microbes can also adhere to the mucosal barrier that covers the intestinal lining. In order to evaluate this fraction of bacteria, ProDigest offers a Mucus-SHIME® or M-SHIME® model with an added mucosal compartment to simulate the intestinal mucus layer.
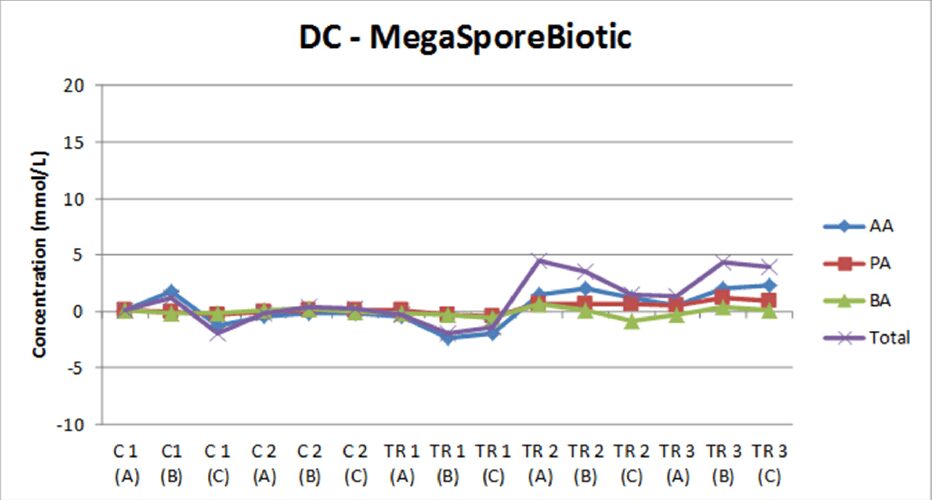
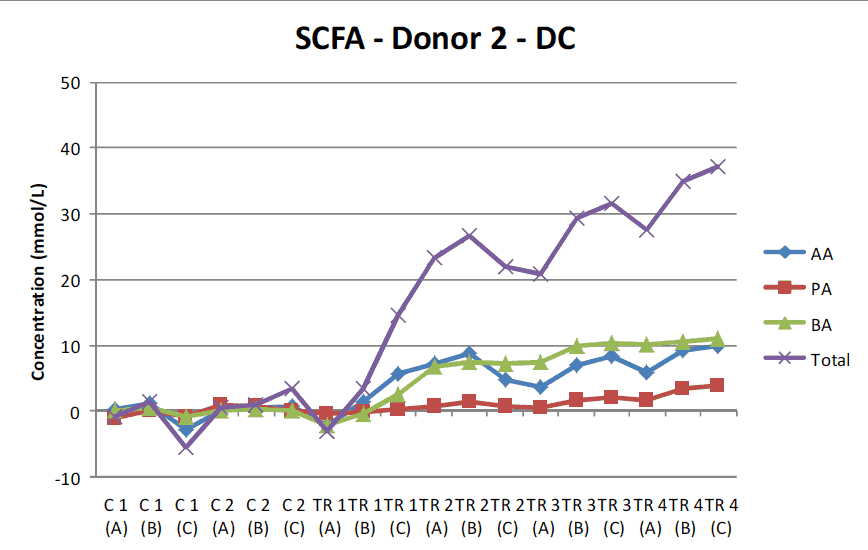
In this symbiotic study, fecal samples were collected from 3 healthy adult subjects, and 3 separate SHIME® experiments were conducted in parallel with the same study design. This particular study included a 2-week initiation period in order to establish a stable microbial community, followed by a 2-week control period and a 4-week treatment period during which the symbiotic test product was administered once daily. At the end of the 8 weeks, researchers concluded that the combination of MegaSporeBiotic and MegaPreBiotic significantly increased microbial diversity in the transverse and descending colon with specific increases in the abundance of Bifidobacterium spp, Lactobacillus spp, Faecalibacterium prausnitzii, and Akkermansia muciniphila, which is representative of a healthy gut microbiome.18 Additionally, this prebiotic/probiotic combination provided significant increases in the production of all short-chain fatty acids (SCFAs) within the gut, including acetate, propionate, and butyrate.
When testing MegaSporeBiotic alone, the SHIME® model revealed a 24-70% increase in total SCFAs among donors. However, this latest study published in The International Journal of Pharmaceutics: X shows that taking MegaPreBiotic with MegaSporeBiotic not only increases microbial diversity in the gut, but this combination also increased total SCFA production by 80-140% among donors.18 For this reason, it is highly recommended to take MegaPreBiotic in conjunction with MegaSporeBiotic.
MegaPrebiotic Indications
Leaky Gut
Autoimmune Disorders
Seasonal Allergies
Food Allergies
Food Sensitivities
Irritable Bowel Syndrome
Crohn’s Disease
Ulcerative Colitis
Diverticulitis
Celiac Disease
Candida Overgrowth
Parasitic Infection
Clostridium difficile Infection
Helicobacter pylori Infection
Small Intestinal Bacterial Overgrowth (SIBO)*
Small Intestinal Fungal Overgrowth (SIFO)
Acne
Psoriasis
Eczema
Diabetes
Obesity
Infertility
Polycystic Ovarian Syndrome
Metabolic Syndrome
Cardiovascular Disease
Kidney Disease
Liver Disease
Alzheimer’s Disease
Parkinson’s Disease
Hepatic Encephalopathy
Recent Antibiotic Use
* may not be tolerated by everyone with SIBO
MegaPrebiotic Advantages
The advantage of using a precision prebiotic over a generalized prebiotic is that you can selectively target your beneficial bacteria without also feeding an unwanted overgrowth of problematic bacteria. MegaPreBiotic contains a much smaller dose of prebiotic fibers than most products on the market which makes it easier to tolerate and less likely to produce gas and bloating. Additionally, these particular fibers have been shown to significantly increased important keystone strains of bacteria in the gut responsible for producing butyrate, modulating fat metabolism, and decreasing intestinal inflammation.
Furthermore, as previously mentioned in the symbiotic study, MegaPreBiotic significantly enhances SCFA production and microbial diversity when taken with MegaSporeBiotic. This powerful combination promotes intestinal repair, microbial balance, and a much healthier immune system. Rather than simply pouring more gasoline on the fire, this dynamic duo can help to first RECONDITION the gut and crowd out any problematic bacteria while encouraging the beneficial bacteria to thrive.
Suggested Use of MegaPrebiotic
Mix with 16 oz of cold water or blend into your favorite smoothie. Start by taking ½ scoop daily for 1 week, then increase to 1 scoop daily in the evening, or as directed by your physician. MegaPreBiotic is safe for children and women who are pregnant or nursing. MegaPreBiotic does not need to be taken with food or at any specific time of day. MegaPreBiotic can be mixed with MegaMucosa and taken simultaneously, but this combination is not necessary. If MegaPreBiotic is too sweet, you can dilute the solution by adding more water. This drink mixture can also be frozen to make popsicles.
MegaPrebiotic Synergy
MegaPreBiotic is most effective when taken with the other products in the Total Gut Restoration system: MegaSporeBiotic (to RECONDITION the gut) and MegaMucosa (to REBUILD the protective mucosal barrier in the gut). As previously mentioned, MegaPreBiotic significantly enhances the beneficial effects of MegaSpore by REINFORCING those beneficial changes to the gut. Taking MegaPreBiotic with MegaSporeBiotic will significantly increase microbial diversity and the production of SCFAs in the gut, which are necessary for healthy intestinal barriers and immunity. Nourish the powerful ecosystem in your gut with MegaPreBiotic today!
MegaPrebiotic Duration
MegaPreBiotic will be most effective when taken daily on the Total Gut Restoration system. The duration of this system will vary by the individual and symptoms but should generally last for 3-6 months. After that point, you can continue taking MegaPreBiotic daily or drop down to a maintenance dose by taking it once a week.
MegaPrebiotic Contraindications
Some patients with SIBO can tolerate oligosaccharides better than others. Following a low FODMAP diet may be helpful in the short-term, but this diet really only serves as a band-aid for SIBO symptoms and can have detrimental effects on the gut microbiome in the long-term. If you have SIBO, start slow with MegaPreBiotic. If you notice an increase in gas or bloating, or worsening of other SIBO symptoms, simply hold off MegaPreBiotic until after you have repaired the intestinal mucosal barrier with MegaMucosa.
If you do have SIBO, it is best to start with MegaSporeBiotic and MegaGuard to RECONDITION the gut microbiome and improve gut motility.
References
- Azcarate-Peril MA, et al. Impact of short-chain galactooligosaccharides on the gut microbiome of lactose-intolerant individuals. Proc Natl Acad Sci USA. 2017;114(3):E367-375.
- Naito Y, et al. A next-generation beneficial microbe: Akkermansia muciniphila. J Clin Biochem Nurt. 2018;63(1):33-35.
- Dao MC, et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. 2016;65:426-436.
- Martin R, et al. Functional Characterization of Novel Faecalibacterium prausnitzii Strains Isolated from Healthy Volunteers: A Step Forward in the Use of F. prausnitzii as a Next-Generation Probiotic. Front Microbiol. 2017; 8: 1226.
- Picard C, et al. Review article: bifidobacteria as probiotic agents – physiological effects and clinical benefits. Aliment Pharmacol Ther. 2005;22:495–512.
- Stenman LK, et al. Potential probiotic Bifidobacterium animalis ssp. lactis 420 prevents weight gain and glucose intolerance in diet-induced obese mice. Benef Microbes. 2014;5(4):437-445.
- Collado MC, et al. Imbalances in fecal and duodenal Bifidobacterium species composition in active and non-active celiac disease. BMC Microbiol. 2008;8:232.
- Gao X, et al. Obesity in school-aged children and its correlation with gut E. coli and Bifidobacteria: a case-control study. BMC Pediatr. 2015;15:64.
- Akay HK, et al. The relationship between bifidobacterial and allergic asthma and/or allergic dermatitis: a prospective study of 0-3-year-old children in Turkey. Anaerobe. 2014;28:98-103.
- Blatchford P, Stoklosinski H, Eady S, et al. Consumption of kiwifruit capsules increases Faecalibacterium prausnitzii abundance in functionally constipated individuals: a randomised controlled human trial. J Nutr Sci. 2017; 6: e52.
- Everard A, Lazarevic V, Derrien M, et al. Responses of Gut Microbiota and Glucose and Lipid Metabolism to Prebiotics in Genetic Obese and Diet-Induced Leptin-Resistant Mice. Diabetes. 2011 Nov; 60(11): 2775–2786.
- Finegold SM, et al. Xylooligosaccharide increases bifidobacteria but not lactobacilli in human gut microbiota. Food Funct. 2014;5(3):436-45.
- Depeint F, et al. Prebiotic evaluation of a novel galactooligosaccharide mixture produced by the enzymatic activity of Bifidobacterium bifidum NCIMB 41171, in healthy humans: a randomized, double-blind, crossover, placebo-controlled intervention study 1–3. Am J Clin Nutr. 2008;87(3):785-91.
- Silk DB, et al. Clinical trial: the effects of a trans-galactooligosaccharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment Pharmacol Ther. 2009;29(5):508-18.
- Vulevic J, et al. Effect of a prebiotic galactooligosaccharide mixture (B-GOS®) on gastrointestinal symptoms in adults selected from a general population who suffer with bloating, abdominal pain, or flatulence. Neurogastroenterol Motil. 2018;30(11):e13440.
- Huaman JW, et al. Effects of Prebiotics vs a Diet Low in FODMAPs in Patients With Functional Gut Disorders. Gastroenterology. 2018;155(4):1004-1007.
- Mego M, et al. Metabolic adaptation of colonic microbiota to galactooligosaccharides: a proof-of-concept-study. Aliment Pharmacol Ther. 2017;45(5):670-680.
Gut Microbiome & Digestion Products
-
-
-
-
$119.98
ZenBiome™ Sleep is formulated to help clients deal with occasional...
Get Social
Recent Podcasts
Recent Courses
Toxicity & Detoxification Masterclass 2024
The toxicity and Detoxification Masterclass covers a wide
Brain & Nervous System Masterclass 2024
19 Leading Experts Share Cutting-Edge Science, Effective Practices,
Autoimmune Masterclass 2024
Autoimmune Masterclass brings together 17 of the world’s
Toxicity & Detoxification Masterclass
The toxicity and Detoxification Masterclass covers a wide
Get the RHT Newsletter
Be the first to get access to special offers, new podcasts, courses, products and events from Rebel Health Tribe.
Facebook
Twitter
Pinterest
LinkedIn
Reddit
WhatsApp
Telegram
StumbleUpon
-
HistaHarmony™
$46.99If you get fatigued, flushed, or foggy after...
-

- Select options This product has multiple variants. The options may be chosen on the product page
MegaSporeBiotic
$59.99 – $176.95MegaSporeBiotic™ is a 100% spore-based, broad-spectrum probiotic shown...
-
MegaSporeBiotic For Kids Gummies
$39.00MegaSporeBiotic™ Gummies are a 100% spore-based proprietary probiotic...
-
MegaMucosa
$59.99MegaMucosa is the first complete mucosal support supplement...

Meet Your Microbiome: The Human Microbiome and How to Optimize It
Meet Your Microbiome: The Human Microbiome and How to Optimize It On and in the average human, there reside approximately

Benefits of Probiotics Including Improved Gut Health
Benefits of Probiotics Including Improved Gut Health “Probiotics” has become quite the buzzword – with countless products and supplements proudly

Gut Microbiome Health and the Gut-Immune System Connection: Part 2 Lower GI
Gut Microbiome Health and the Gut-Immune System Connection: Part 2 Lower GI The diverse community of microbes in your digestive
Gut Microbiome Health and the Gut-Immune System Connection: Part 1 Upper GI
Gut Microbiome Health and the Gut-Immune System Connection: Part 1 Upper GI The message is out: gut health is critical




















